- HealthTech and Pharmacy
- SaaS
- Business Processes Automation
Pharmaceutical Regulatory Records Management System
A custom SaaS regulatory records management system built for pharmaceutical teams that need to centralize product, territory, authorization, and documentation data. The platform supports controlled record workflows, lifecycle tracking, secure file management, role-based access, auditability, and notifications for important regulatory dates.

Overview
Quick overview: key aspects of our work - discover the essentials of our project
Industry
Country
Solution type
Services
About the client
Understanding our client: specifics, challenges, and custom solutions
The client is a life sciences technology company that develops software solutions for pharmaceutical and healthcare workflows. Their focus covers regulatory affairs, quality assurance, document management, manufacturing process support, electronic batch records, and training management.
Problem
Solution
Key features
Project features overview: essential enhancements and strategic solutions
-
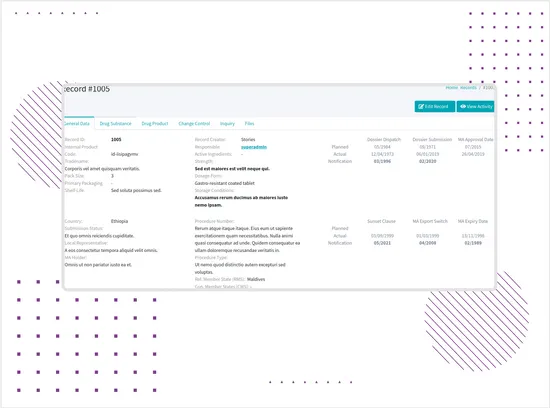
Complex Regulatory Record Forms
Structured forms were built to manage detailed pharmaceutical product records, including territory-specific data, authorization stages, manufacturer and supplier details, product attributes, dossier dates, GMP dates, and documentation fields.The form logic helps teams keep regulatory data consistent, organized, and easier to update throughout the product lifecycle. -
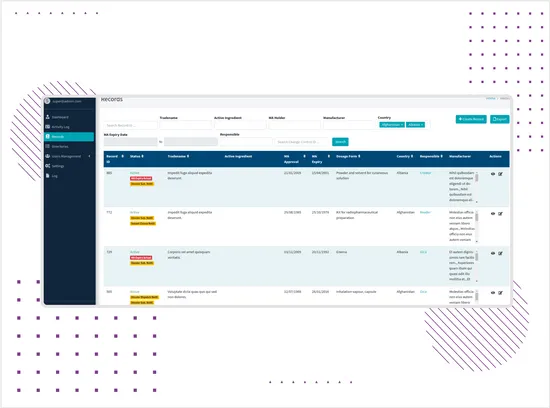
Fast Search and Filtering
The platform includes search, filtering, sorting, and table-based navigation to help users find records by key regulatory fields such as Record ID, tradename, active ingredient, marketing authorization holder, manufacturer, country, and expiry date.This gives teams faster access to product and territory-specific information without manually checking scattered files or spreadsheets. -

Flexible Export
Authorized users can export structured record data for reporting, review, and operational planning. Exports can be based on selected fields, filtered records, and predefined report types such as monthly reports, change control progress reports, and renewal plans.This makes regulatory data easier to prepare, share, and review outside the platform while keeping the original record structure intact. -
Built-In Secure File Storage and File Management
The system includes file storage connected directly to regulatory records. Users can upload, organize, preview, download, rename, move, and export files within a controlled record-based structure.This helps keep documentation linked to the relevant product, country, authorization, manufacturer, supplier, or regulatory process instead of being stored separately from the record context. -
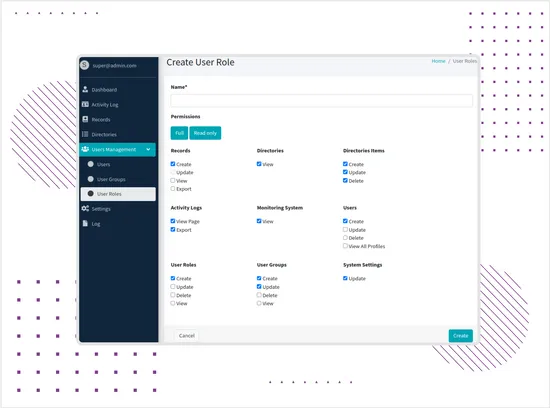
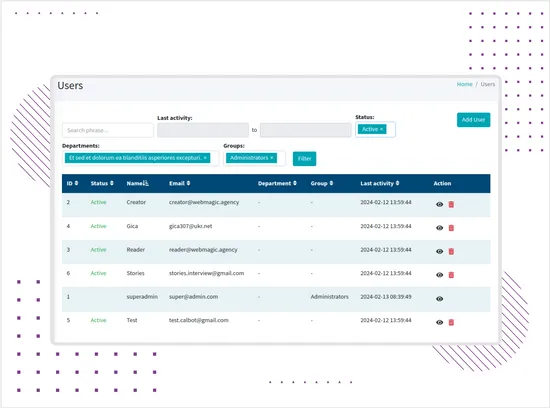
Role-Based Permissions and Access Control
The platform supports multi-level access management based on users, roles, groups, departments, record creators, and responsible users. Permissions control access to records, exports, directories, user management, system settings, and activity logs.This allows different teams to work in the same system while limiting access to sensitive data and actions based on responsibility. -
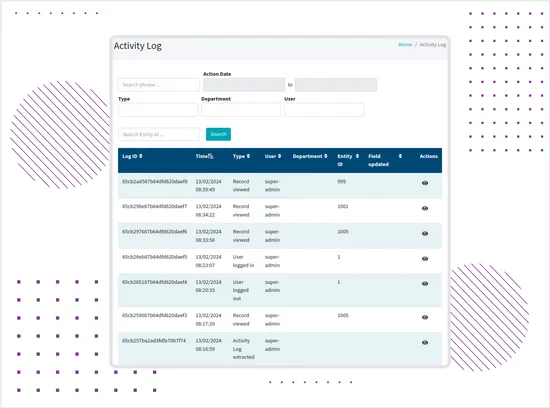
User Activity Logs
The system records important user actions, including access attempts, record-related actions, permission-restricted events, and administrative activity.These logs support traceability and make it easier for authorized users to review what happened in the system, who performed an action, and when it occurred. -
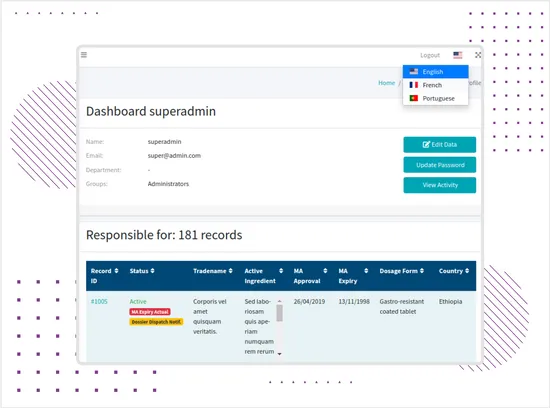
Multilingual Data Entry Support
The system supports data entry in different languages where free-text fields are used, while keeping the core interface structure consistent for operational clarity.This helps teams manage international regulatory records that may include country-specific product data, documentation details, local market information, and multilingual source materials.
Result
Performance Showcase: Unveiling the Results of Our Collaborative Endeavors
Customer review
Real Stories of Satisfaction and Success
Do you have a business challenge you’d like to resolve?
If you have an idea or a problem that you would like to eliminate in your business processes, leave a request. We will be happy to discuss this with you at a free consultation and find the most suitable solution for your specific situation
Discover how WebMagic transforms ideas into intelligent digital solutions. Our case studies highlight real projects in AI-powered automation, logistics platforms, SaaS tools, e-commerce systems, and custom web development.
From streamlining supply chains to building smart dashboards and integrating wearable data with AI — each case shows how we solve complex problems with scalable, tech-driven systems.
We work with clients across industries to deliver high-impact results: faster processes, clearer insights, and stronger digital infrastructure.
Explore how we’ve helped startups, enterprises, and growing teams turn challenges into success — and imagine what we can build for your business.